- HOME
- VENUE
- RSVP
- REGISTRY
- CONTACT
- Bytefence anti malware pro
- Tomb raider yamatai monastary
- Canon mg3100 scanner not working
- Average speed equation
- Outlook express 6 2-2-1 serial key
- Ps4 s-pdif port
- Root apk 4-4-4 download
- Excel standard deviation
- Daemon tools safe
- Ps1 controller vs ps4 co
- D3dcompiler_43-dll windows 10
- Ios rom emulator
- Command and conquer red alert 3 ps4
- Price check prince of persia 3d 90s
- With fire and sword cheats
- Free download ancient aliens all seasons
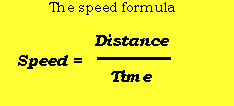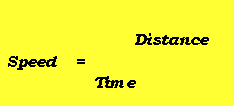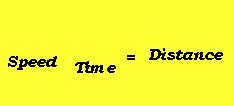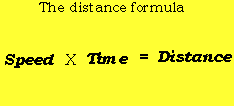
Effect of Particle Mass on Average SpeedĬonsider the equation for kinetic energy,įrom this equation, it is clear that for particles with the same kinetic energy, as mass increases, the velocity would decrease by a power of two. The different lines represent normal distributions at different temperatures, in Celsius.įrom this graph, it is clear that as temperature increases from -100 to 600, the amount of particles with higher kinetic energy increases. In this graph, known as a Maxwell Botlzmann distribution, n represents the fraction of particles, and v represents the kinetic energy of the particle. By plotting these normal distributions together, we can see that as temperature increases, the amount of particles experiencing higher speeds increases, and if temperature decreases, the amount of particles experiencing higher speeds decreases. However, a sample of gas actually has a range of speeds, forming a normal distribution with a different height and width for different temperatures. Because of this, you might assume that all particles would have the same speed. It states that all individual particles are moving in random directions with random speeds.įrom our 5th assumption, recall that this random velocity is a function of absolute temperature.
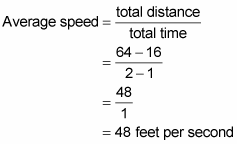
Kinetic molecular theory of gases is a way to describe the random motion of particles in a sample of gas. So what is the kinetic molecular theory definition? This means that as temperature decreases to absolute zero, particle kinetic energy and speed decrease to zero as well.The kinetic energy of gas molecules is proportional to the absolute temperature only.This means that when two gas particles collide, kinetic energy is conserved during the collision.Collisions between gas particles are completely elastic.Gas particles are in motion and move randomly in one direction until they collide with another body.We also must make assumptions regarding motion of particles, i.e. Gas particles exert no attractive or repulsive forces on other gas particles.The volume occupied by a single gas particle is negligible in comparison to the volume occupied by the gas itself.Average speed of gas particles can be calculated with the root-mean-square formula.įor kinetic molecular theory, we use the basic assumptions for ideal gases like helium, which are.Recall that velocity is a vector, with both direction and magnitude, whereas speed is a scalar, with only magnitude.If particles are moving with equal probability in different directions, the average velocity will be 0.All gas particles move in random directions with random speeds.
AVERAGE SPEED EQUATION HOW TO
You will learn the kinetic molecular theory of gases, and how to use the root-mean-square formula to determine the average speed of gas.